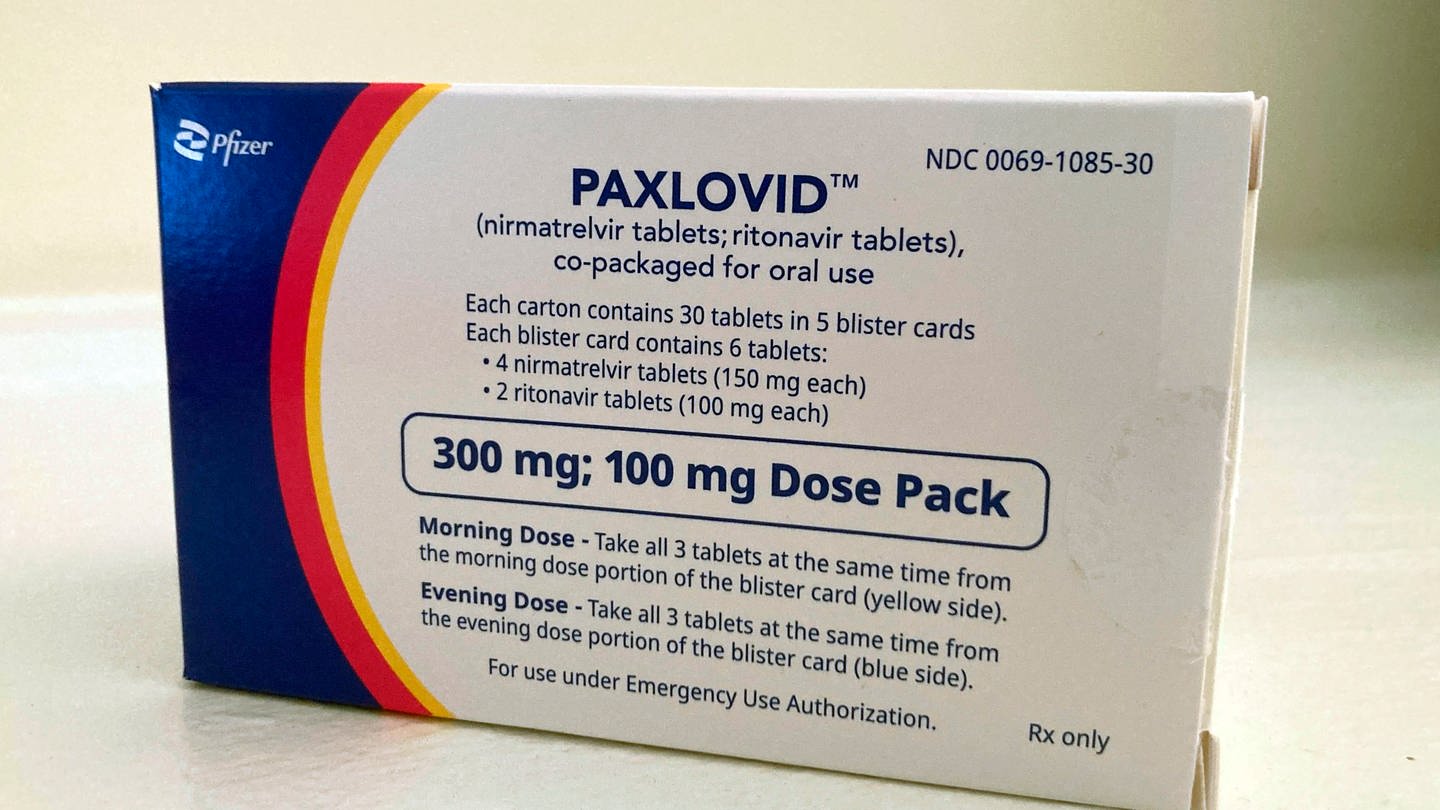
Paxlovid
National Center for Biotechnology Information. It found taking Paxlovid orally within the first five days of infection was tied to a 25 relative decrease in developing 10 of the 12 most common long COVID symptoms including shortness of.

Rki Covriin
Due to the potential for severe drug-drug interactions with the ritonavir component of Paxlovid it is strongly suggested that healthcare providers not experienced in prescribing this drug refer to.

. About Paxlovid Who can and cannot take it How and when to take it Side effects Pregnancy breastfeeding and fertility Taking Paxlovid with other medicines and herbal supplements. Past studies have shown that Paxlovid reduces the risks of hospitalization and death from COVID-19. PAXLOVID is not an appropriate therapeutic option based on the authorized Fact Sheet for Healthcare Providers or due to potential drug interactions for which recommended monitoring would not be feasible.
What Prescribers and Pharmacists Need to Know. Ontario is considering allowing pharmacists to prescribe the COVID-19 treatment drug Paxlovid in order to expand access the provinces top doctor says. Ritonavir tablets Adverse Event Report.
Paxlovid Find out how Paxlovid treats coronavirus COVID-19 and how to take it. Since the trigger of long COVID is acute infection with SARS-CoV-2 it makes intuitive sense. We have produced various materials in PDF format to aid the use of experimental agents in the treatment of COVID-19.
The study shows the challenge of demonstrating a benefit of antiviral therapy in previously vaccinated or otherwise low risk individuals said Paul Sax clinical director of the Division of Infectious. The new study says that Paxlovid may help stave off some of those issues. In addition the three oral drugs did not increase the occurrence of adverse events thus.
Call 1-800-232-0233 TTY 1-888-720-7489 to get help in English Spanish and more than 150 other languages8 AM to midnight ET 7 days a week. Studies from the drugmaker showed that in unvaccinated people at risk of serious COVID. In late April a study found that Paxlovid wasnt successful as a preventive measure for people who had been exposed to the virus.
Call the Centers for Disease Control and Prevention CDC COVID-19 hotline which can provide information on the Test to Treat initiative. Since Paxlovid must be taken within five days after symptoms begin authorizing state-licensed pharmacists to prescribe Paxlovid could expand access to timely treatment for some patients who. Improving patient care through guidelines evidence summaries and decision aids that we can all trust use and share.
These resources are freely available and the Liverpool Drug Interactions Group would like to encourage the dissemination with an appropriate acknowledgement of this drug-drug interaction information for non-commercial use. Paxlovid a five-day course of pills from Pfizer is at the top of the list of recommended treatments. National Center for Biotechnology Information.
Paxlovid is for individuals age 12 and older who have tested positive for COVID-19 are at increased risk of severe disease and have developed COVID-19 symptoms in the last 5 days even mild ones such as runny nose or cough. 6 2022 Read the full story here. KQED provides public radio television and independent reporting on issues that matter to the Bay Area.
Were the NPR and PBS member station for Northern California. In this study we used population-based real world data to evaluate the effectiveness of Paxlovid. Paxlovid was granted emergency use authorization for the treatment of mild to moderate COVID-19 based on the interim analysis of EPIC-HR trial.
Paxlovid eligibility refer to FDAs Fact Sheet for Healthcare Providers. 1For information on medical conditions and factors associated with increased risk for progression to severe COVID-19. One rising variant BA2752 is particularly concerning for its potential to thwart the last monoclonal antibody left in the COVID arsenal.
Researchers found the decreased risk of long COVID associated with the medication exists regardless of whether it was a. Also known as PAXLOVID nirmatrelvir tablets. Paxlovid is also contraindicated with drugs that conversely strongly induce those same enzymes leading to the faster breakdown of nirmatrelvir or ritonavir as reduced concentrations of.
Whether you are a patient caregiver or healthcare professional it is important to report adverse events for Pfizer products. Ontario COVID-19 Drugs and Biologics Clinical Practice Guidelines Working Group on behalf of the Ontario COVID-19 Science Advisory Table. Paxlovid effectiveness needs to be assessed in a noncontrolled setting.
This study showed that three novel oral antivirals molnupiravir fluvoxamine and Paxlovid are effective in reducing the mortality and hospitalization rates in patients with COVID-19. Learn how FDA prepares for and responds to emergencies and how FDA plays a role in protecting the United States from chemical biological radiological nuclear and emerging infectious disease.

Paxlovid For A Patient On A Doac Ontario Covid 19 Science Advisory Table

Covid Medikament Bund Kauft Eine Million Einheiten Paxlovid Tagesschau De

Paxlovid Hhs Aspr
Lauterbach Pusht Paxlovid Anti Corona Mittel Fur Risikogruppen Aber Gefahrlich Focus Online
As More People Report Covid Rebounds After Paxlovid Experts Insist Cases Are Rare

Paxlovid Molnupiravir Und Co Was Die Corona Medikamente Konnen Br24

Paxlovid Lagevrio Lagerhaltung Erlaubt Apotheke Adhoc

Ema Empfiehlt Covid 19 Medikament Paxlovid

Paxlovid Schon Bei Leichtem Corona Ptaheute

Paxlovid Als Hoffnungstrager Vorteile Und Nachteile Des Covid Medikaments Swr2
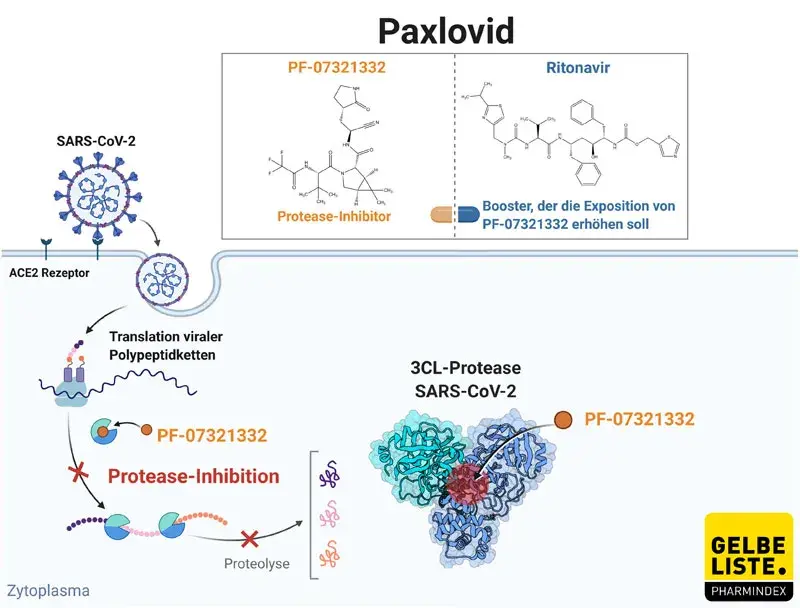
Paxlovid Konnte Neuer Blockbuster Der Corona Behandlung Werden Gelbe Liste

Paxlovid Cuts Risk Of Long Covid Study Finds Everyday Health
Paxlovid S Slow Targeted Rollout Leaves Vulnerable Populations At Risk News Northeastern
)
Us Making It Easier To Access Covid Treatment Paxlovid Community Healthcare System

Pfizer S Paxlovid Anti Viral Seen To Lower Long Covid Risk In Study The Japan Times

For Some Covid Patients Paxlovid Rebound Has Nothing To Do With Paxlovid Los Angeles Times
Corona Medikament Kaum Verschrieben Dr Barbara Romer Paxlovid Is Kein Medikament Der Masse Swr1